2023-05-30
Humans have lost half of primate ancestors' gut bacteria, finds new study
(Summary)
현존 인류는 고대 조상에 비해 장내 미생물의 절반을 상실
새로운 연구에 따르면 영장류의 장내에는 수백만 년 동안 많은 박테리아 그룹이 진화해 왔지만, 인간은 이러한 공생 박테리아의 절반 가까이를 잃었습니다. 인간 장내 미생물 군집의 이러한 변화의 원인은 밝혀지지 않았지만, 이 연구의 저자들은 식단의 변화가 이러한 차이를 초래한 것으로 추정하고 있습니다. 멸종된 박테리아의 차이는 마이크로바이옴을 변화시키는 것으로 알려진 항생제와 같은 현대식 식단 및 의약품과 관련된 차이일 수 있습니다. 일부 연구자들은 이러한 장내 미생물의 파괴가 자가 면역 질환이나 대사 증후군과 같은 현대 질병에 영향을 미칠 수 있다고 추측하고 있습니다.
 Humans have lost half of primate ancestors' gut bacteria, finds new study
Humans have lost half of primate ancestors' gut bacteria, finds new study
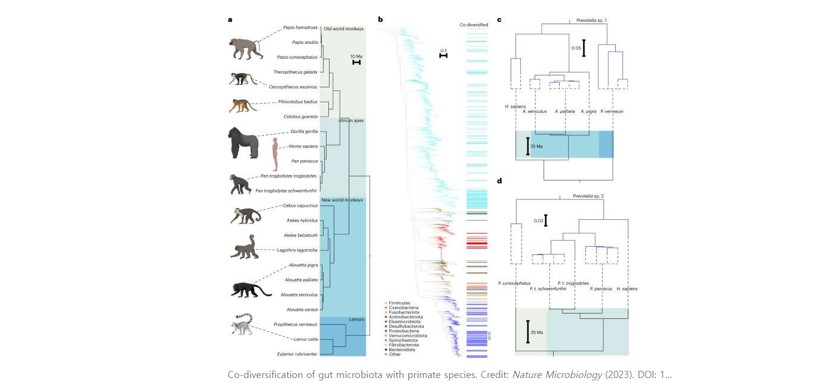
A new study finds that hundreds of bacterial groups have evolved in the guts of primate species over millions of years, but humans have lost close to half of these symbiotic bacteria.
In the study, researchers compared populations of gut bacteria found in chimpanzees and bonobos, our closest relatives, with those of humans—which in total amount to some 10,000 different lineages of bacteria. The scientists analyzed the evolutionary relationships of these bacteria in primates and identified groups of bacteria that were present in distant ancestors of humans and primates. Strikingly, the results showed that these ancestral symbionts are being lost rapidly from the human lineage.
Though the cause of these shifts in human gut microbiomes is not known, the study's authors suspect changing diets probably caused the divergence.
"The working idea is that the losses we see spanning all human populations, regardless of lifestyle, were likely driven by dietary shifts that happened early in human evolution since we've diverged from chimpanzees and bonobos," said Andrew Moeller, assistant professor of ecology and evolutionary biology and faculty curator of mammalogy at the Cornell University Museum of Vertebrates, in the College of Agriculture and Life Sciences, and the paper's senior author.
In particular, human diets shifted away from complex plant polysaccharides found in leaves and fruits towards more animal fat and protein, Moeller said.
Jon Sanders, a former postdoctoral researcher in Moeller's lab, is first author of the study, "Widespread Extinctions of Co-diversified Primate Gut Bacterial Symbionts From Humans," published May 11 in Nature Microbiology. Daniel Sprockett, a current postdoctoral researcher in Moeller's lab, is also a co-author.
In the study, the researchers analyzed metagenomes, which are assembled by piecing together short base pair sequences from a whole community of genomes; the metagenomes revealed which microorganisms were present in a sample and their relative abundances.
Analyses of 9,640 human and non-human primate metagenomes, including newly generated ones from chimpanzees and bonobos, revealed significant evidence that gut bacteria groups shared an evolutionary history with their hosts, according to the paper.
The results showed that 44% of clades—a group that has evolved from a common ancestor—that have a shared evolutionary history with African apes were absent from the human metagenomic data and 54% were absent from industrialized human populations. At the same time, only 3% of bacterial clades in African apes that did not share an evolutionary history with these hosts were absent in humans.
"This is the first microbiome-wide study showing that there are a great number of ancestral co-diversifying [shared evolution] bacteria that have been co-living within primates and humans for millions of years," Moeller said.
Still, Moeller highlighted the importance of improved sampling in human populations, especially those outside of industrial countries, in order to fully represent human gut microbiome diversity.
Ancestral bacteria may be passed from one generation to another from mothers to babies, and by social transmission with other members of the same species.
The discrepancy in extinct bacteria between the general human population and those from industrialized countries may point to differences related to modern diets and medicines, such as antibiotics that are known to alter microbiomes. Some researchers have speculated that the disruption of ancestral flora could be playing a role in modern diseases, such as autoimmune disorders and metabolic syndrome.
A second related paper, "Home-site Advantage for Host Species-specific Gut Microbiota," also led by Moeller and Sprockett, which published May 12 in Science Advances, showed that gut bacteria locally adapt to the hosts they live in, providing a possible mechanism for the long-term stability of these symbioses.
Provided by Cornell University
More Information
https://phys.org/news/2023-05-humans-lost-primate-ancestors-gut.html
현존 인류는 고대 조상에 비해 장내 미생물의 절반을 상실
새로운 연구에 따르면 영장류의 장내에는 수백만 년 동안 많은 박테리아 그룹이 진화해 왔지만, 인간은 이러한 공생 박테리아의 절반 가까이를 잃었습니다. 인간 장내 미생물 군집의 이러한 변화의 원인은 밝혀지지 않았지만, 이 연구의 저자들은 식단의 변화가 이러한 차이를 초래한 것으로 추정하고 있습니다. 멸종된 박테리아의 차이는 마이크로바이옴을 변화시키는 것으로 알려진 항생제와 같은 현대식 식단 및 의약품과 관련된 차이일 수 있습니다. 일부 연구자들은 이러한 장내 미생물의 파괴가 자가 면역 질환이나 대사 증후군과 같은 현대 질병에 영향을 미칠 수 있다고 추측하고 있습니다.
 Humans have lost half of primate ancestors' gut bacteria, finds new study
Humans have lost half of primate ancestors' gut bacteria, finds new studyA new study finds that hundreds of bacterial groups have evolved in the guts of primate species over millions of years, but humans have lost close to half of these symbiotic bacteria.
In the study, researchers compared populations of gut bacteria found in chimpanzees and bonobos, our closest relatives, with those of humans—which in total amount to some 10,000 different lineages of bacteria. The scientists analyzed the evolutionary relationships of these bacteria in primates and identified groups of bacteria that were present in distant ancestors of humans and primates. Strikingly, the results showed that these ancestral symbionts are being lost rapidly from the human lineage.
Though the cause of these shifts in human gut microbiomes is not known, the study's authors suspect changing diets probably caused the divergence.
"The working idea is that the losses we see spanning all human populations, regardless of lifestyle, were likely driven by dietary shifts that happened early in human evolution since we've diverged from chimpanzees and bonobos," said Andrew Moeller, assistant professor of ecology and evolutionary biology and faculty curator of mammalogy at the Cornell University Museum of Vertebrates, in the College of Agriculture and Life Sciences, and the paper's senior author.
In particular, human diets shifted away from complex plant polysaccharides found in leaves and fruits towards more animal fat and protein, Moeller said.
Jon Sanders, a former postdoctoral researcher in Moeller's lab, is first author of the study, "Widespread Extinctions of Co-diversified Primate Gut Bacterial Symbionts From Humans," published May 11 in Nature Microbiology. Daniel Sprockett, a current postdoctoral researcher in Moeller's lab, is also a co-author.
In the study, the researchers analyzed metagenomes, which are assembled by piecing together short base pair sequences from a whole community of genomes; the metagenomes revealed which microorganisms were present in a sample and their relative abundances.
Analyses of 9,640 human and non-human primate metagenomes, including newly generated ones from chimpanzees and bonobos, revealed significant evidence that gut bacteria groups shared an evolutionary history with their hosts, according to the paper.
The results showed that 44% of clades—a group that has evolved from a common ancestor—that have a shared evolutionary history with African apes were absent from the human metagenomic data and 54% were absent from industrialized human populations. At the same time, only 3% of bacterial clades in African apes that did not share an evolutionary history with these hosts were absent in humans.
"This is the first microbiome-wide study showing that there are a great number of ancestral co-diversifying [shared evolution] bacteria that have been co-living within primates and humans for millions of years," Moeller said.
Still, Moeller highlighted the importance of improved sampling in human populations, especially those outside of industrial countries, in order to fully represent human gut microbiome diversity.
Ancestral bacteria may be passed from one generation to another from mothers to babies, and by social transmission with other members of the same species.
The discrepancy in extinct bacteria between the general human population and those from industrialized countries may point to differences related to modern diets and medicines, such as antibiotics that are known to alter microbiomes. Some researchers have speculated that the disruption of ancestral flora could be playing a role in modern diseases, such as autoimmune disorders and metabolic syndrome.
A second related paper, "Home-site Advantage for Host Species-specific Gut Microbiota," also led by Moeller and Sprockett, which published May 12 in Science Advances, showed that gut bacteria locally adapt to the hosts they live in, providing a possible mechanism for the long-term stability of these symbioses.
Provided by Cornell University
More Information
https://phys.org/news/2023-05-humans-lost-primate-ancestors-gut.html

